39 isotopes and average atomic mass worksheet
PDF Isotopes and average atomic masses worksheet Isotopes and average atomic masses worksheet answers. The element bromine has three isotopes naturally appealing. A mass spectrum of molecular BR2 shows three peaks with mass numbers of 158 u, 160 u, and 162 u. Use this information to determine which Bri isotopes occur in nature. Ã 79 U, 81 mg Calculates the elementary atomic mass if the ... PDF Isotope Practice Worksheet 9. Iodine is 80% 127I, 17% 126I, and 3% 128I. Calculate the average atomic mass of iodine. 10. The natural abundance for boron isotopes is 19.9% 10B and 80.1% 11B . Calculate boron's atomic mass. 11. Hydrogen is 99% 1H, 0.8% 2H, and 0.2% 3H. Calculate its average atomic mass. 12. Rubidium is a soft, silvery-white metal that has two common ...
PDF Chem 11 Worksheet 8-1: Name Isotopes & Average Atomic Mass ... Answer: .750 x 133 = 99.75 .200 x 132 = 26.4 .050 x 134 = 6.70 . Total = 132.9 a.m.u. = average atomic mass Determine the average atomic mass of the following mixtures of isotopes. 1. 80.0% 127 53I , 17.0% 126 53I , 3.0% 128 53I 1. 2. 50.0% 197 79Au , 50.0% 198 79Au 2.
Isotopes and average atomic mass worksheet
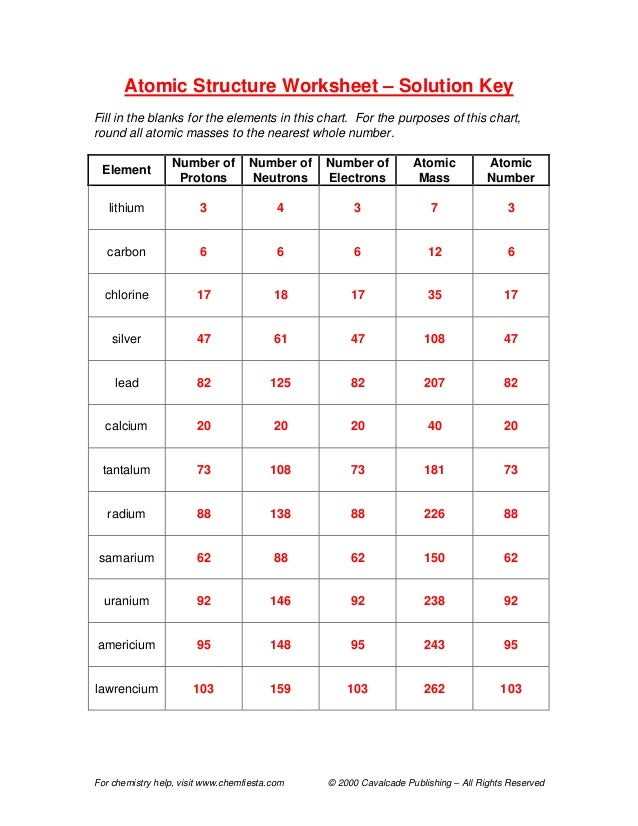
PDF Isotope Practice Worksheet - Chemistry 7. Boron exists in two isotopes, boron-10 and boron-11. Based on the atomic mass, which isotope should be more abundant? Answer: The atomic mass of boron is 10.811; therefore, boron-11 is more abundant because the mass number is closer to the atomic mass. 8. Lithium-6 is 4% abundant and lithium-7 is 96% abundant. What is the average mass of ... avg atomic mass WS KEY.pdf The average atomic mass between these two isotopes is 63.546 amu. Calculate the actual atomic mass of Cu. (62.9298Y016909) + ()(0.3091) - 63.546. 1431478 ... DOC Calculating Average Atomic Mass Worksheet Name Calculate the average atomic mass of sulfur if 95.00% of all sulfur atoms have a mass of 32, 0.76% has a mass of 33 and 4.22% have a mass of 34. 3. The four isotopes of lead are shown below, each with its percent by mass abundance and the composition of its nucleus.
Isotopes and average atomic mass worksheet. Quiz & Worksheet - Isotopes and Average Atomic Mass ... Isotopes and average atomic mass, as concepts, allow for the specific discussion of elements and their atoms, and this quiz/worksheet combo will help you test your understanding of these concepts. Calculating Average Atomic Mass Worksheet - CHEM 33000 ... Calculating Average Atomic Mass Worksheet. Show ALL calculation setups. The term "average atomic mass" is a __weighted_____average, and so is calculated differently, from a "normal" average. Explain how this type of average is calculated. This average is calculated by summing masses of the element's isotopes. PDF Atoms and Isotopes Worksheet - Tumwater School District Atoms and Isotopes Worksheet 1. Fill in the table with the correct information. Isotope Isotope Notation Atomic # Protons Electrons Neutrons Oxygen-16 Bromine-80 Uranium- 235 Copper-64 2. Describe the general arrangement of subatomic particles in the atom. 3. What contribution did these scientists make to atomic models of the atom? a. Calculating Average Atomic Mass Worksheet Name - Magoffin ... The element copper has naturally occurring isotopes with mass numbers of 63 and 65. The relative abundance and atomic masses are: 69.2% for mass of 62.93u. 30.8 ...
PDF Isotopes and Average Atomic Mass - FISD One isotope, which has a percent abundance of 72.15% has a mass of 84.9118 amu. The other isotope has a mass of 86.9092 amu. a. Calculate the average atomic mass of this element to three decimal places Isotope Atomic Mass % Abundance 1 84.9118 ∗72.15/100 = 61.2638637 2 86.9092 ∗27.85/100 = + 24.2042122 85.4680758 ANS. DOC Isotope Practice Worksheet - University of Manitoba The natural abundance for boron isotopes is 19.9% 10B (10.013 amu*) and 80.1% 11B (11.009 amu*). Calculate the atomic mass of boron. Average atomic mass = [(19.9%)(10.013)] + [(80.1%)(11.009)] 100 = 10.811 (note that this is the value of atomic mass given on the periodic table) PDF Average Atomic Mass Problems Key 2013 Chem Isotopes and Average Atomic Mass Page 9/22. Online Library Average Atomic Mass Problems Key 2013 Average Atomic Mass Problems Key Calculate the average atomic mass of an element with the follow isotope information: 4.35% have a mass of 49.9461 amu, 83.79% have amass of 51.9405 amu, 9.50% have a mass PDF Calculating Average Atomic Mass Worksheet - NMSU Calculating Average Atomic Mass Worksheet: 1) Three isotopes of Silicon occur in nature: Atomic Mass: 27.97693 amu 28.97649 amu 29.97377 amu Isoto es of Silicon: Silicon-28 Silicon-29 Silicon-30 Percent Abundance: 92.23% 4.68% 3.09% Calculate the average atomic mass for the three isotopes of Silicon. (-9 Y(.o ( zq.q 7377) 0/0 2) Two isotopes Of ...
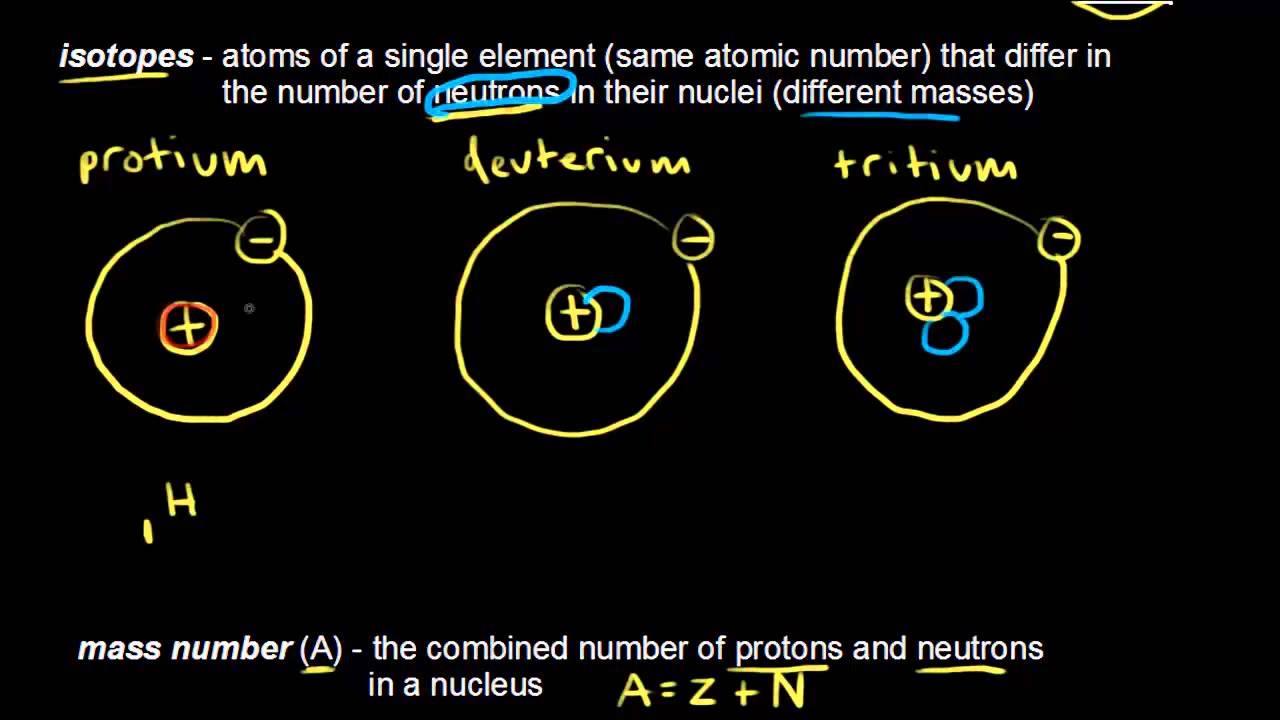
PDF Isotopes Average Atomic Mass - nyostrander.us Isotopes and Average Atomic Mass Isotopes are atoms of the same element (they have the same number of protons) but with different numbers of neutrons. The atomic mass reported in the Periodic Table is the weighted average of the naturally occurring isotopes. Determine the weighted average atomic mass of the following elements. 1. Isotopes And Average Atomic Mass Worksheets - Kiddy Math Isotopes And Average Atomic Mass - Displaying top 8 worksheets found for this concept.. Some of the worksheets for this concept are Chm 4 pal atomic mass student name, Livingston public schools lps home, Livingston public schools lps home, Honors chem atomic, Mayfield high school, Abundance of isotopes chemistry work 43 answer key, Isotopes and mass spectrometry, Unit 3. Worksheet #4 – Isotopes & Average Atomic Mass Answer the following questions regarding average atomic mass and isotopes. Show all work for problems involving calculations! Define the word “isotope”. True or ... PDF Answers Key for Unit Worksheets - Livingston The average atomic mass between these two isotopes is 63.546 amu. Calculate the actual atomic mass of 65Cu. X — amð 7) Magnesium consists of three naturally occurring isotopes. The percent abundance of these isotopes is as follows: 24 Mg (78.70%), 25Mg (10.13%), and 26Mg(11.7%). 'The average atomic mass of the three isotopes is 24.3050 amu.
11 Best Images of Atom Worksheets With Answer Keys - Atoms Ions and Isotopes Worksheet Answer ...
PDF Isotopes and Average Atomic Mass This is done the same way you would find any weighted average: Calculating the Atomic Mass 1) Convert given percent abundances to decimals (divide by 100) 2) Multiply the mass of each isotope by its decimal abundance. 3) Add resulting numbers. Ex 1) Find the average atomic mass of carbon given the following: Isotope Mass (amu) Percent Abundance
Isotopes and Atomic Mass - Isotopes | Atomic Mass - PhET ... Give evidence to support or dispute: "In nature, the chance of finding one isotope of an element is the same for all isotopes.". Find the average atomic mass of an element given the abundance and mass of its isotopes. Predict how the mass and name of an isotope will change given a change in the number of protons, neutrons or electrons.

Average Atomic Mass and Percent Abundance Worksheet 2 and KEY | Isotope | Chemical Elements ...
PDF Atomic Mass Average Atomic Mass Overview The average atomic mass of an element can be determined from the relative amounts of each isotope. This is the mass used in most chemical calculations. In a naturally occurring element, the fractional abundance is the percentage of the abundance of a particular isotope in the total sample of atoms, written as a decimal.
PDF isotopic abundance practice problems - CHEMISTRY The three silicon isotopes have atomic masses and relative abundances of 27.9769 amu (92.2297%), 28.9765 amu (4.6832%) and 29.9738 amu (3.0872%). ! 4. Gallium has two naturally occurring isotopes. The mass of gallium-69 is 68.9256 amu and it is 60.108% abundant. The mass of gallium-71 is 70.9247 amu and it is 39.892% abundant.
PDF NAME Average Atomic Mass Worksheet: show all work. The average atomic mass of the three isotopes is 24.3050 amu. If the atomic mass of 25Mg is 24.98584 amu, and 26Mg is 25.98259 amu, calculate the actual atomic mass of 24Mg. 24Mg = 23.98504 amu 8) Complete the table Isotope Mass (amu) Relative Abundance (%) Neon-20 19.992 90.51 Neon-21 20.994 0.27 Neon-22 21.991 9.22
Isotopes Worksheet - Google Docs 3. Naturally occurring europium (Eu) consists of two isotopes was a mass of 151 and 153. Europium-151 has an abundance of 48.03% and Europium-153 has an abundance of 51.97%. What is the average atomic mass of europium? 4. Strontium consists of four isotopes with masses of 84 (abundance 0.50%), 86 (abundance of 9.9%), ...
PDF Isotope Worksheet Answer Key - ISD 622 Determine the average atomic mass of the following mixtures of isotopes. 128 127 126 a. t, 17%- 3% I (sðf' 8) 197 198 19 q. 5 55 56 Fe, 85% 55) 55.85 12 14 (03115) GoaYlG) 13,3 + (H 5) + ( , 3 a) 2. How many neutrons does Zn-66 have? mass 3. Give the hyphen notation for this atom, p=76, e= 76, n= 116 11, is 11 on Is (93, This is 4.
Isotopes And Average Atomic Mass Worksheet Answers Isotopes And Average Atomic Mass Worksheet Answers. 19 May 2021 Added forthcoming change to active mode appraisal toolkit. The list of databases and different objects refreshes automatically when the worksheet context is changed. Users also can click on the refresh button at the high of the object browser to view object modifications immediately.
Worksheet Mass Relative Atomic [W6ZN2V] Search: Relative Atomic Mass Worksheet
PDF KMBT 654-20131024112244 - Berger's Chemistry Class What is its average atomic mass? Answer: .75 x 133 = 99.75 .20 x 132 = 26.4 .05 x 134 = Total = 132.85 amu = average atomic mass Determine the average atomic mass of the following mixtures of isotopes. l. IZC 2. 500/0 3. 150/0 4. Il-I, 0.8%4-1, 0.2% 0.01b 5. OMS 13, B 0t46 6. 980/0 Chemistry IF87ó6
27 Chemistry Atomic Number And Mass Number Worksheet Answers - Notutahituq Worksheet Information
Calculating Average Atomic Mass Worksheet: Calculating Average Atomic Mass Worksheet: 1) Three isotopes of Silicon occur in nature: Isotopes of Silicon: Percent Abundance: Atomic Mass: Silicon-28 92.23% 27.97693 amu Silicon-29 4.68% 28.97649 amu Silicon-30 3.09% 29.97377 amu Calculate the average atomic mass for the three isotopes of Silicon.
Review Mass Average Worksheet Atomic [G90TM2] Search: Average Atomic Mass Review Worksheet
DOC Calculating Average Atomic Mass Worksheet Name Calculate the average atomic mass of sulfur if 95.00% of all sulfur atoms have a mass of 32, 0.76% has a mass of 33 and 4.22% have a mass of 34. 3. The four isotopes of lead are shown below, each with its percent by mass abundance and the composition of its nucleus.
avg atomic mass WS KEY.pdf The average atomic mass between these two isotopes is 63.546 amu. Calculate the actual atomic mass of Cu. (62.9298Y016909) + ()(0.3091) - 63.546. 1431478 ...







0 Response to "39 isotopes and average atomic mass worksheet"
Post a Comment